Research
The Chemistry Department is located in the Merkert Chemistry Center, a 109,000 square foot structure designed to empower the dynamic, cutting-edge research in organic chemistry, chemical biology, and physical chemistry for which the department is internationally recognized.
Latest Research
Research Archive
02.27.2023 Udayan Mohanty and Jia Niu: “Clickable Disaccharides” Lead to Efficient Assembly of Glycomimetics
12.28.2022 Jia Niu and Dunwei Wang: Photocatalytic Lignin Valorization
05.20.2022 Udayan Mohanty: Diffuse Ions Coordinate Dynamics in a Ribonucleoprotein Assembly
01.18.2022 Jianmin Gao: Lysine-Targeting Reversible Covalent Inhibitors with Long Residence Time
08.27.2021 Jia Niu: Metathesis Cascade-Triggered Depolymerization of Enyne Self-Immolative Polymers
04.14.2021 Dunwei Wang and Matthias Waegele: Artificial Photosynthesis
04.08.2021 Peter Zhang: Metalloradical Activation of Carbonyl Azides for Enantioselective Radical Aziridination
11.25.2020 Peter Zhang: Enantioselective Intermolecular Radical C−H Amination
11.11.2020 Udayan Mohanty:A steric gate controls P/E hybrid-state formation of tRNA on the ribosome
07.09.2020 Jia Niu: A General Approach to O‐Sulfation by a Sulfur(VI) Fluoride Exchange Reaction
06.08.2020 Shih-Yuan Liu: Pd-Senphos Catalyzed trans-Selective Cyanoboration of 1,3-Enynes
09.12.2019 Udayan Mohanty: Cooperative relaxation in deeply supercooled liquids
07.24.2019 Lawrence T. Scott (Emeritus): Negatively-curved Warped Nanographene Self-Assembled on Metal Surfaces
07.15.2019 Dunwei Wang: Electrochemical CO2 reduction
10.22.2018 Peter Zhang: Catalytic Radical Process for Enantioselective Amination of C(sp3)‐H Bonds
07.25.2018 Dunwei Wang: New binding mode of heterogeneous catalysts discovered
06.18.2018 Jia Niu: Radical Cascade-Triggered Controlled Ring-Opening Polymerization of Macrocyclic Monomers
05.02.2018 Dunwei Wang & Matthias Waegele: Selective CO production by Photoelectrochemical Methane Oxidation
04.12.2018 Dunwei Wang: Cathodically Stable Li-O2 Battery Operations Using Water-in-Salt Electrolyte
03.22.2018 James Morken: Synthesis and Stereochemical Assignment of Arenolide
03.07.2018 Dunwei Wang: "New heterogeneous catalysts for solar water splitting"
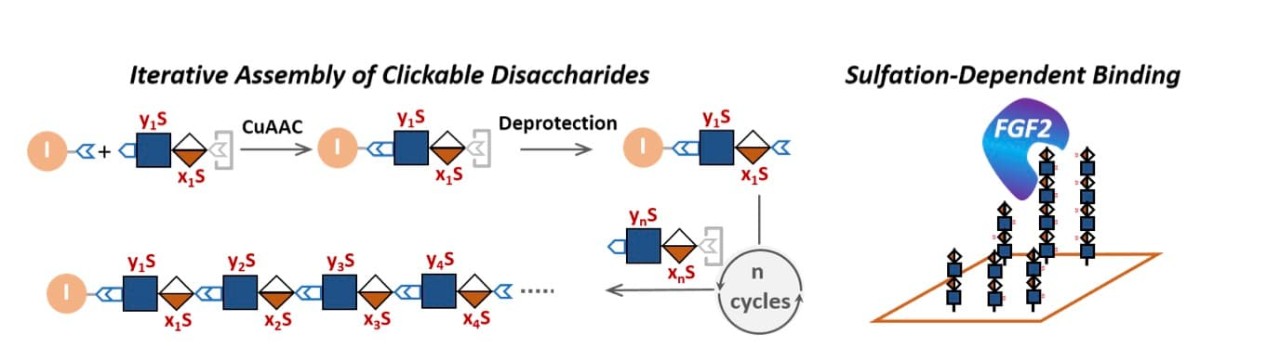
“Clickable Disaccharides” Lead to Efficient Assembly of Glycomimetics
Udayan Mohanty and Jia Niu
February 27, 2023
Cell surface glycans play important roles in a variety of cell functions ranging from signaling transduction to cell-pathogen interactions to cancer development. However, understanding of the structures and functions of cell surface glycans has been hampered by their highly heterogenous structure and modifications. Recently Professors Jia Niu and Udayan Mohanty, postdoc Cangjie Yang, and graduate students Yu Deng and Yang Wang teamed up to tackle this challenge. They developed a facile approach to generating glycomimetics with complex sulfation patterns through iterative assembly of clickable disaccharide building blocks that mimic the disaccharide repeating units of heparan sulfate, a type of highly sulfated cell surface glycan. The team demonstrated that assembly of variably sulfated clickable disaccharides can facilely generate a library of mass spec-sequenceable glycomimetic oligomers with defined sulfation patterns by solution-phase iterative syntheses. Molecular dynamics (MD) simulations further confirmed that these glycomimetic oligomers bind protein fibroblast growth factor 2 (FGF2) in a sulfation-dependent manner consistent with that of the native glycans. This work established a general approach to glycomimetics that can potentially serve as alternatives to native glycans in both fundamental research and disease models.
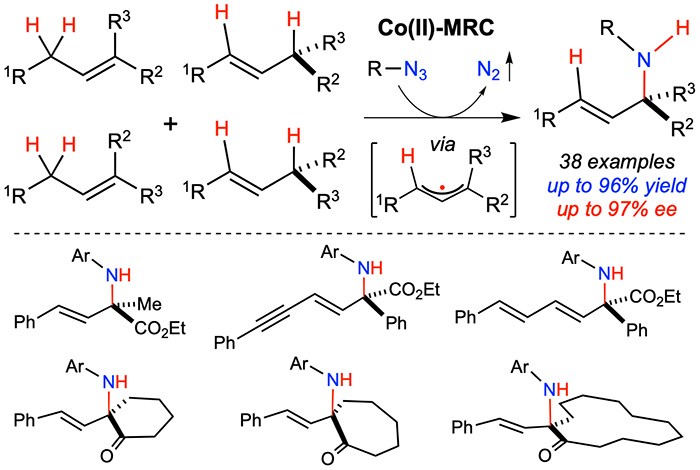
Homolytic radical chemistry has a number of inherent features for bond-breaking and bond-forming in chemical synthesis that are distinctly different from heterolytic ionic chemistry. During the past decades, radical reactions have been increasingly explored as new synthetic tools for construction of molecular structures. The outcome of these growing efforts may impact the planning and practice in organic synthesis. While radical chemistry offers vast potentials, the control of reactivity and selectivity stands as the major roadblock toward synthetic application of many radical reactions. Besides the differentiation of multiple competing pathways that typically coexist in a radical process, controlling various selectivities in radical reactions presents formidable challenges. This is well illustrated in the textbook Wohl-Ziegler reaction for radical functionalization of allylic C–H bonds involving the delocalized allylic radicals as the intermediates. This classic organic free radical reaction, which typically generates a regioisomeric mixture of products, faces the long-standing challenge in controlling multi-selectivities such as chemoselectivity and regioselectivity as well as diastereoselectivity and enantioselectivity. Recently, Professor Peter Zhang and coworkers report a metalloradical approach for concurrent control of multiple convergences and selectivities in intermolecular radical allylic C–H amination via Co(II)-based metalloradical catalysis (MRC). The Co(II)-based metalloradical system enables chemoselective intermolecular amination of allylic C−H bonds with remarkable convergence of regioselectivity as well as diastereoselectivity and enantioselectivity in one single catalytic operation. It offers a route to synthesize valuable chiral α-tertiary amines directly from an isomeric mixture of alkenes.
Read the article in Nature Chemistry.
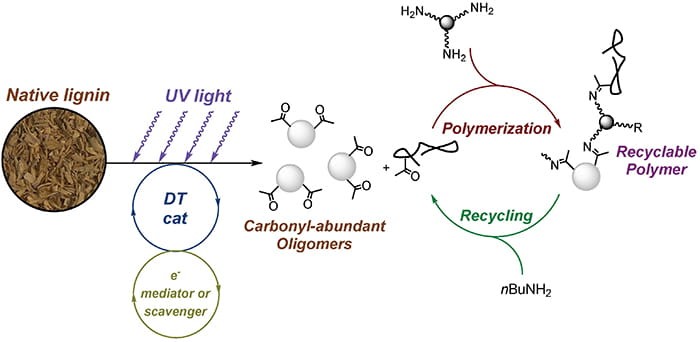
Lignin is a nonedible biomass resource that is rich in chemical functionalities. It holds great promise for sustainable chemical synthesis. To date, however, the chemical utilization of lignin has been limited. This is because the existing biomass valorization processes prioritize cellulose and hemicellulose and induce significant condensation reactions to the labile bonds in lignin, making it exceeding difficult to decompose lignin for subsequent chemical synthesis. Moreover, in the handful examples where lignin can be decomposed, the resulting materials are a mixture of complex compositions that are prohibitively expensive to separate. Recently, Professors Wang and Niu teamed up to tackle this challenge. They found that it is possible to selectively depolymerize native lignin under mild conditions by a photocatalytic approach, so as to minimize condensation reactions to the labile linkages. Importantly, they show that the degree of depolymerization can be controlled for the production of oligomers with desired molecular weight and chemical functionality, which can be readily utilized for the synthesis of covalent dynamic polymer networks. The resulting materials are easy to recycle and represent an example of circular economy.
Read the article in ACS Central Science.
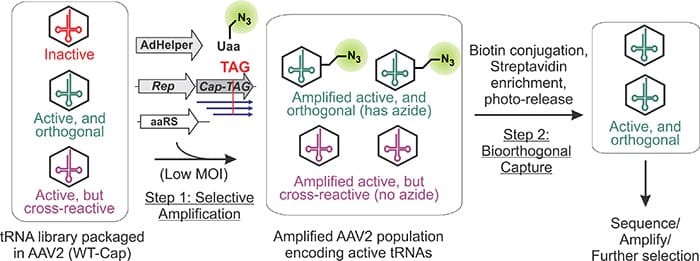
Directed evolution is a revolutionary technology that allows scientists to harness the power of evolution to create biomolecules with novel functions. Although directed evolution has been used with much success in simple unicellular organisms like bacteria and yeast, extending it to humancells has remained difficult. Inspired by the ability of viruses to evolve within human cells, the Chatterjee group has harnessed this ability to develop a virus-assisted directed evolution strategy (VADER) in mammalian cells. In this paper, published in Nature Methods, the authors demonstrate that by linking the activity of a tRNA to the replication of a human virus (adeno-associated virus), it is possible to evolve mutant tRNAs with improved activities in human cells.
Read the article in Nature Methods.
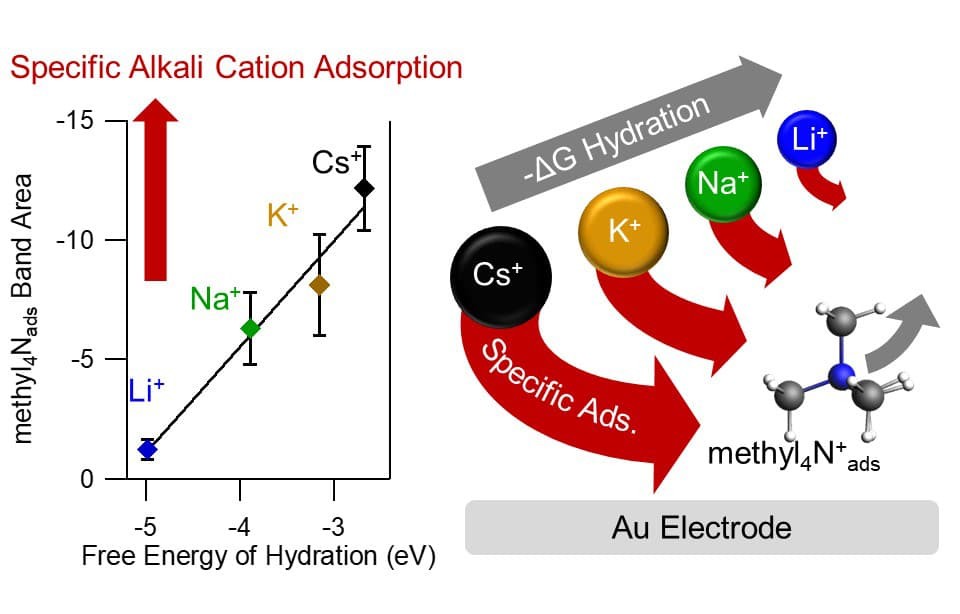
Alkali metal cations of the supporting electrolyte can greatly influence the rates and product selectivity of reactions occurring at electrocatalytic interfaces. Alkali metal cations in direct contact with the electrode, known as specifically adsorbed cations, have been hypothesized to play a central role. However, their detection remains elusive. In their latest advance, Prof. Waegele’s team, in collaboration with Prof. Michael Janik at Penn State, developed tetramethylammonium as a vibrational reporter for the distribution of alkali metal cations at the aqueous Au interface. Using this approach, they found spectroscopic evidence for specifically adsorbed alkali metal cations during CO2-to-CO conversion. Importantly, they determined that the propensity of alkali metal cations to specifically adsorb on Au correlates with their free energy of hydration. That is, the softer the hydration shell of an alkali metal cation, the higher its surface coverage for a given bulk concentration. The study further shows that the rate of CO formation increases with the amount of specifically adsorbed cations on the electrode. The findings indicate that partial dehydration of alkali metal cations plays a key role in promoting CO2-to-CO reduction. These insights are expected to aid the design of electrocatalytic interfaces with high catalytic activity and desirable product selectivity.
Read the article in Nature Catalysis.
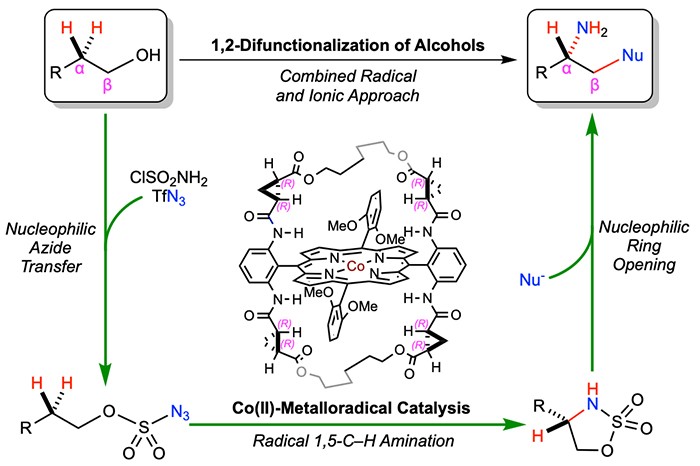
The past decade has seen enormous efforts in developing either radical or ionic approaches to access chiral molecules. Among different research targets, chiral amines represent one of the most important classes of organic compounds with widespread applications in biology, medicine, and materials. Enantioselective synthesis of chiral amines is considered to be one of the cornerstones in stereoselective organic synthesis. To this end, the direct functionalization of omnipresent C–H bonds with common organic nitrogen compounds is among the most appealing methods. However, C–H amination strategies are largely limited to constructing a specific type of N-heterocycles or amine derivatives. To maximize the synthetic potential of asymmetric C–H amination, Professor Peter Zhang and coworkers envision an attractive approach that unites the complementary reactivities of radical and ionic chemistry for the streamlined synthesis of functionalized chiral amines. They report a general approach for the stereoselective synthesis of α-chiral amines bearing various β-functionalities from readily available alcohols through successfully merging the development of a new enantioselective radical process for C(sp3)–H amination with an enantiospecific ionic process for nucleophilic ring-opening of the resulting amination product. In addition to demonstrating site-selectivity, stereospecificity, and nucleophile diversity for the ionic ring-opening process, they have developed the first catalytic radical process for enantioselective 1,5-C(sp3)–H amination of alcohol-derived alkoxysulfonyl azides via Co(II)-based metalloradical catalysis (MRC). To showcased the potency of this combined radical and ionic approach, they have stereoselectively constructed both linear and cyclic molecular structures bearing multiple contiguous stereocenters and functionalities that would be normally difficult or impossible to access through existing methods.
Read the article in Nature Synthesis.
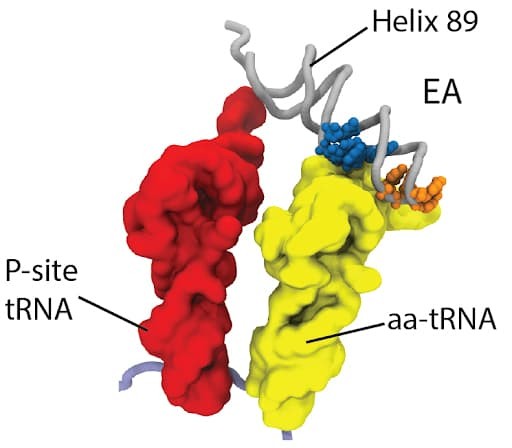
It is well known that ions are essential to the dynamics of biomolecular assemblies, especially those that contain nucleic acids. The precise physical-chemical contributions of loosely-associated diffuse ions to large-scale biomolecular dynamics remains an outstanding challenge. To enable the quantitative investigation of the effects of diffuse ions in large-scale biomolecular assemblies, a team led by Udayan Mohanty, Ailun Wang, Paul C. Whitford and Mariana Levi, developed large-scale molecular dynamics simulations techniques that explicitly describe all-atom (non-H) resolution and explicit ions to show that the model accurately predicts experimentally reported distributions of diffuse ions for prototypical RNA systems. We demonstrate the utility of this approach by simulating the model to a complete ribosome in the presence of physiologically relevant concentrations of Mg2+, K+ and Cl- ions. This uncovers a direct relationship between diffuse ions and a conformational shift in an extended region of the assembly. Further, we find that diffuse ions facilitate long-range attraction and recruitment of tRNA by the ribosome.
Read the article in Journal of the American Chemical Society.
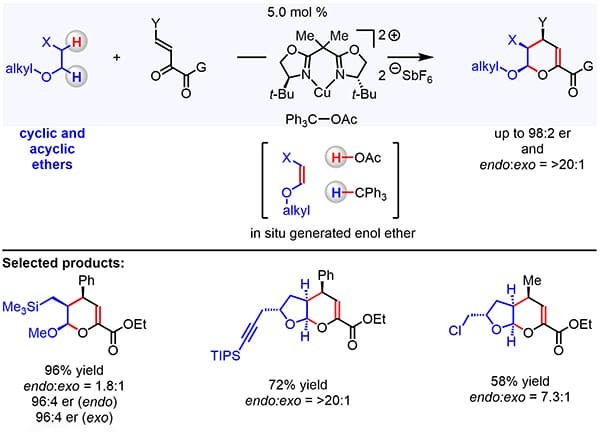
Wasa and co-workers developed an enantio- and diastereo-selective method for the transformations of vicinal C–H bonds within various acyclic and cyclic ethers to generate dihydro-2H-pyran derivatives. By using a blend of chiral organocopper catalyst and Ph3COAc, Wasa’s team discovered that it is possible to convert alkyl ethers into enol ethers and then promote their stereoselective reaction with β,γ-unsaturated ketoesters. The catalyst system is tolerant of a variety of Lewis acid-sensitive functional units and allows for rapid access to valuable chiral ether products containing stereogenic centers at the C1, C2, and C3 positions. These findings demonstrate that the proper combination of a chiral Lewis acid and an in situ generated hydride acceptor may be used for chemo- and enantio-selective functionalization of otherwise stable ether-based molecules, thus providing a rational basis for the future development of methods for the stereoselective synthesis of biologically relevant ether-based molecules, as well as their late-stage functionalization.
Read the article in Journal of the American Chemical Society.
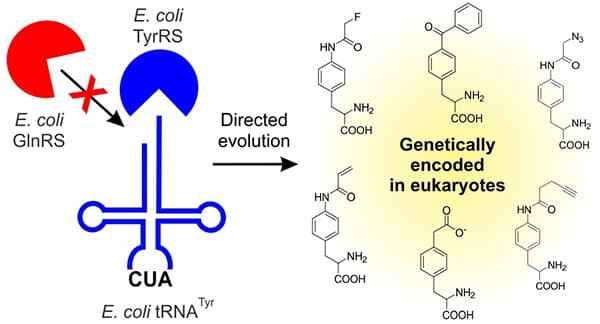
The ability to site-specifically incorporate novel unnatural amino acids (UAAs) into proteins in living cells provides numerous ways to probe and engineer protein function. However, access to UAAs with diverse structural features, which can be incorporated into proteins expressed in eukaryotic cells, have traditionally been restricted due to the difficulty of developing the necessary genetic machinery. To overcome this limitation, the Chatterjee group has developed a method to readily engineer the bacteria-derived tyrosyl-tRNA synthetase/tRNA for incorporating many novel and useful UAAs into eukaryotic proteins. These UAAs enable numerous applications, such as site-specific labeling of any recombinant proteins, and covalently capturing protein-protein interactions through photo-crosslinking or chemical crosslinking. The group also incorporated p-carboxymethyl-phenylalanine, which mimics the post-translational modification phosphotyrosine. This will allow scientists to study the effect of tyrosine phosphorylation, an important post-translational modification of eukaryotic proteins, with unprecedented versatility.
Read the article in ACS Central Science.
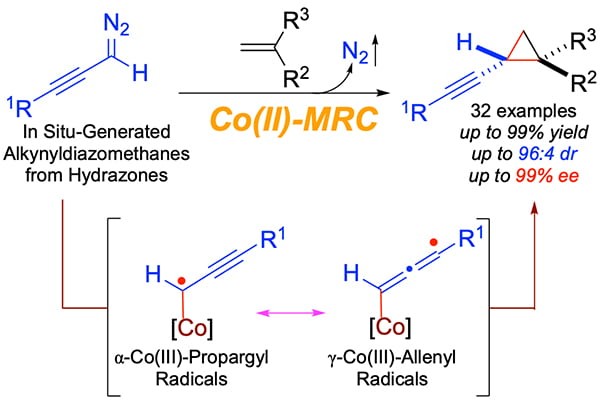
Chiral alkynyl cyclopropanes occur as common substructures in many important natural products and drug molecules. In addition, chiral alkynyl cyclopropanes may serve as useful intermediates for stereoselective organic synthesis. Nevertheless, general and effective methods for their asymmetric synthesis remain to be developed. To meet the synthetic challenge for constructing this class of valuable molecules, Professor Peter Zhang and coworkers demonstrate a metalloradical approach to homolytically activate in situ-generated α-alkynyldiazomethanes for asymmetric radical cyclopropanation of alkenes. They report in this new manuscript the successful development of Co(II)-based metalloradical system that is highly effective for asymmetric radical cyclopropanation of alkenes with α-alkynyldiazomethanes through in situ-generation from the corresponding sulfonyl hydrazones in the presence of base. Through fine-tuning of the D2-symmetric chiral amidoporphyrin ligand, the optimized Co(II)-based catalytic system can efficiently activate different α-alkynyldiazomethanes at room temperature for highly asymmetric cyclopropanation of a broad range of alkenes. The catalytic radical process provides a general synthetic method for stereoselective construction of chiral alkynyl cyclopropanes in high yields with high both diastereoselectivity and enantioselectivity. They have also presented detailed computational and experimental studies that offer several lines of evidence in support of the underlying stepwise radical mechanism for this Co(II)-catalyzed cyclopropanation involving a unique α-metalloradical key intermediate that is associated with two resonance forms of α-Co(III)-propargyl radical and γ-Co(III)-allenyl radical. To demonstrate the synthetic applications of this new methodology, they have shown that the resulting enantioenriched alkynyl cyclopropanes can serve as useful chiral building blocks for different stereoselective transformations.
Read the article in Journal of the American Chemical Society.
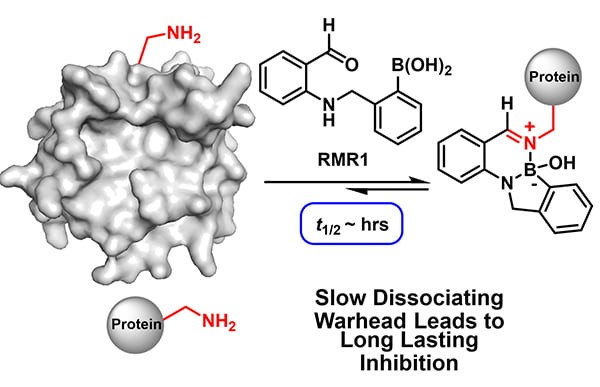
There is much interest in the development of reversible covalent drugs as they can enjoy the benefits of irreversible covalent drugs (high potency and specificity), yet avoid permanent modification of target proteins, which often leads to toxicity. A bottleneck for developing reversible covalent inhibitors lies in the scarcity of reversible reactions that modify important protein residues. In a recent paper published in Journal of the American Chemical Society, Dr. Rahi M. Reja, Professor Jianmin Gao and their coworkers report a novel reversible conjugation reaction for protein lysine side chain amines. Specifically, a designed chemical warhead RMR1 readily conjugates with a lysine side chain amine under physiologic conditions to give a diazaborine product. The diazaborine formation is fully reversible, giving rise to a novel covalent binding mechanism for lysines. Importantly, in comparison to previously known reversible lysine conjugation chemistries, the newly developed diazaborine conjugation exhibits much slowed dissociation kinetics. Taking advantage of this unique reaction profile, the team devised a series of highly effective reversible covalent inhibitors for an important enzyme, sortase of Staphylococcus aureus, which has emerged as an appealing target for anti-Staph therapeutics. Sortase inhibition was demonstrated with recombinant proteins as well as with live S. aureus cells. Detailed mechanistic studies revealed not only the thermodynamic contribution of RMR1 to sortase binding, but also the kinetic benefit of this slow dissociating warhead, which results in long-acting inhibitors.
Read the article in Journal of the American Chemical Society.
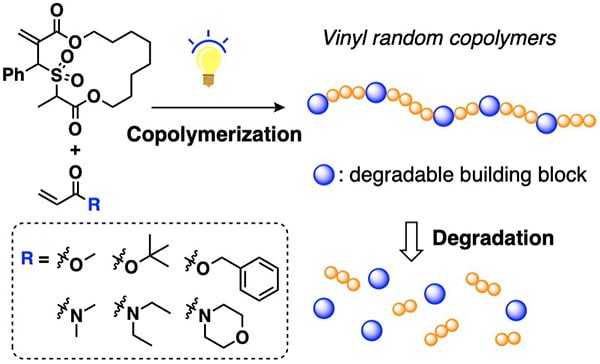
The widespread use of vinyl plastics has revolutionized our society, but also created significant environmental challenges due to their accumulation in the environment. Degradable vinyl polymers are promising solutions to these challenges. However, achieving even distributions of labile functional groups in the backbone of degradable vinyl polymers remains challenging. Writing on Angewandte Chemie International Edition, Professor Jia Niu and graduate students Wenqi Wang and Zefeng Zhou reported a photocatalytic approach to degradable vinyl random copolymers. Based on a technique called radical ring-opening cascade copolymerization, they showed that macrocyclic allylic sulfone monomers containing degradable ester groups can be copolymerized with a variety of acrylate and acrylamide monomers with near-unity reactivity ratios over the entire range of the feed compositions. The resulting vinyl copolymers feature tunable degradable groups evenly distributed in the backbone, allowing for excellent degradability and material properties comparable to the non-degradable vinyl polymers.
Read the article in Angewandte Chemie International Edition.
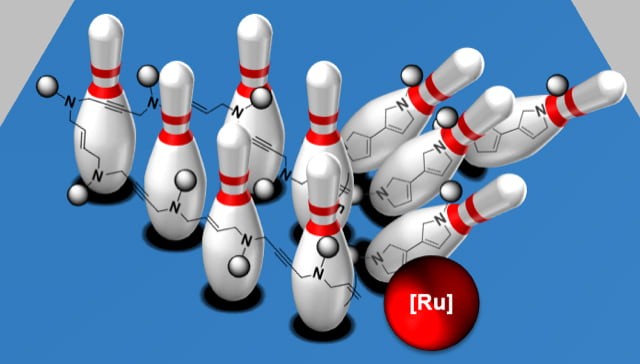
Self-immolative polymers (SIPs) are a promising class of degradable polymers that can undergo triggered head-to-tail depolymerization in the presence of external stimuli. Despite significant advances in the past decade, existing SIPs are still limited by their chemical lability, toxicity, nonoptimal material performance, and inefficient depolymerization. Writing on Angewandte Chemie International Edition, Professor Jia Niu, postdoc Jingsong Yuan, and graduate student Gavin Giardino reported a new strategy to address these challenges. They described a novel class of enyne SIPs that employ rapid and energetically favored olefin metathesis cascade reaction to trigger depolymerization. Model studies revealed a 1,6-enyne motif consisting of a p-pentylbenzenesulfonamide linker enabled efficient metathesis cascade cyclization. The SIPs incorporating this motif were readily prepared via polycondensation and iterative exponential growth approaches. These SIPs were stable in strong acid, base, nucleophiles, or at elevated temperatures, and could undergo efficient and complete depolymerization once triggered by a metathesis catalyst. Temperature-triggered depolymerization of the enyne SIPs was demonstrated using thermally responsive second-generation Grubbs catalyst, highlighting the potential for these enyne SIPs to be used as stimuli-responsive materials. This paper is featured as a Very Important Paper.
Read the article in Angewandte Chemie International Edition.
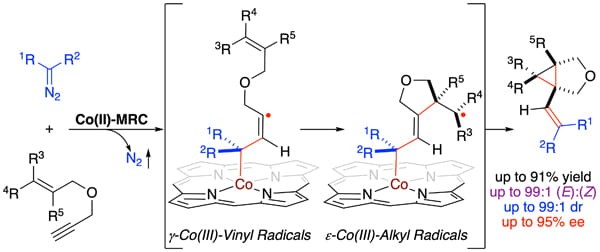
Radical cascade reactions represent attractive synthetic tools for the construction of natural products and complex molecules in organic synthesis. While diastereoselective radical cascade has been successfully applied in many important transformations, control of enantioselectivity remains a formidable challenge in the field. As a new application of metalloradical catalysis (MRC) for addressing the fundamental challenges associated with radical cascade reactions, Professor Peter Zhang and coworkers have disclosed the first asymmetric catalytic system for radical cascade cyclization of 1,6-enynes with diazo compounds that enables the stereoselective construction of 1-alkenylbicyclo[3.1.0]hexane structures, which are prevalent motifs in pharmaceuticals and biologically active compounds. Through fine-tuning of D2-symmetric chiral amidoporphyrins as the supporting ligands, the Co(II)-catalyzed radical cascade system can be effectively applied for the stereoselective synthesis of 1-alkenylbicyclo[3.1.0]hexane structures, which contain multisubstituted cyclopropane-fused tetrahydrofurans bearing three contiguous stereogenic centers, including two all-carbon quaternary centers in high yields with excellent stereoselectivities. Additionally, they presented both computational and experimental studies that shed light on the underlying stepwise radical mechanism for this new cascade bicyclization, which is fundamentally different from the previous catalytic systems involving metallocarbene intermediates. To showcase the synthetic applications of this Co(II)-based radical cascade system, the resulting enantioenriched cyclopropane-fused tetrahydrofurans that contain a tri-substituted vinyl group at the bridgehead can be further utilized for several stereospecific transformations.
Read the article in Journal of the American Chemical Society.
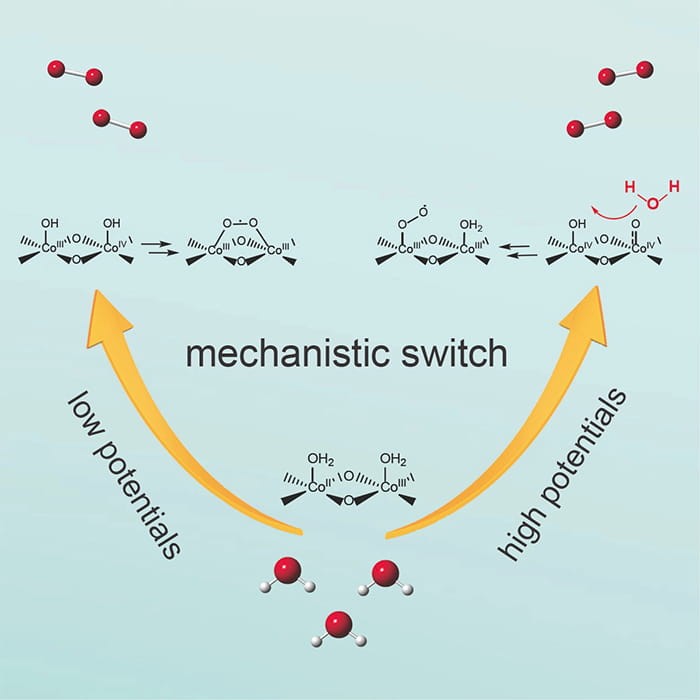
Water oxidation is an important first step of photosynthesis, releasing electrons and protons from water for carbon dioxide and nitrogen fixation reactions and producing diatomic oxygen as a byproduct. Despite decades of intense research, the mechanism of this reaction has remained poorly understood, which is especially true for heterogeneous catalysts that are of vital importance for technological developments. One reason for the challenge is the lack of tools to examine the reaction for a quantitative relationship between the reactants, the catalyst, and the reaction kinetics. Aiming to tackle this problem, Professors Wang and Waegele made an important step forward. In a recent paper published in Chem, the two groups teamed up and made two observations. First, they identified the key reaction intermediates using surface enhanced infra-red spectroscopy. This discovery is important because it clearly proves the chemical route by which diatomic oxygen forms, a critical piece of information in understanding the detailed mechanisms. Second, they observed a mechanistic switch by varying water activities, which allowed them to interrogate how the system responds to changing reactant concentrations. This study represents one of the first to change water concentrations as previous research always treated it as a constant. This approach bypasses the challenge that the concentration of active heterogeneous catalysts is difficult to quantify. It provides important new insight into how water is oxidized. At low driving force, the reaction proceeds through a dinuclear mechanism; when the driving force is high, it switches to a mononuclear one. The results are expected to have major implications in the understanding and improvement of practical water oxidation catalysts. This project is funded by the Solar Photochemistry program in the Basic Energy Science Division of the Department of Energy.
Read the article in Chem.
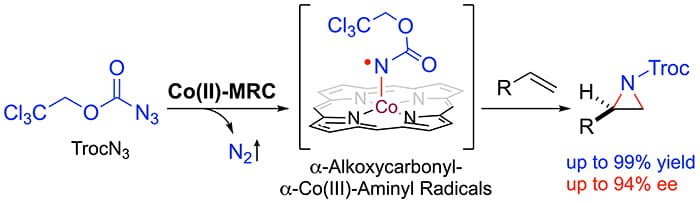
As a new application of metalloradical catalysis (MRC), Professor Peter Zhang and coworkers has recently reported a catalytic radical approach for asymmetric olefin aziridination with the carbonyl azide TrocN3. Among common organic azides, carbonyl azides have not been previously demonstrated as effective nitrogen sources for intermolecular olefin aziridination despite their potential formation of useful aziridines bearing N-carbonyl functionality. This underdevelopment is mainly attributed to the well-known challenges associated with the high lability of carbonyl azides toward thermal and photolytic rearrangements. They describe that cobalt(II) complexes of chiral porphyrins are effective metalloradical catalysts to activate TrocN3 even at room temperature to aziridinate alkenes, providing chiral N-carbonyl aziridines in high yields with excellent enantioselectivities. This represents the first example of asymmetric intermolecular olefin aziridination that employs carbonyl azides as the nitrogen source. In addition to detailed computational and experimental studies on its underlying stepwise radical mechanism, they have demonstrated the synthetic applications of the resulting N-Troc-aziridines as valuable chiral synthons for stereoselective synthesis of other chiral aziridines and various chiral amine derivatives.
Read the article in Chem.
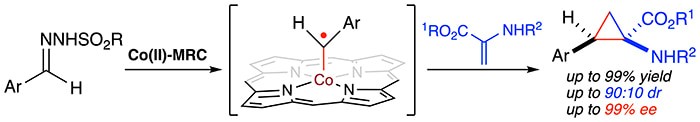
Chiral cyclopropyl α-amino acids represent a unique class of non-proteinogenic α-amino acids that have found wide applications in the design and synthesis of novel peptides with restricted conformations for biological studies. As an exciting new application of metalloradical catalysis (MRC), Professor Peter Zhang and coworkers has reported the successful development of a Co(II)-based metalloradical system that is highly effective for asymmetric cyclopropanation of dehydroaminocarboxylates with in situ-generated α-aryldiazomethanes. Supported by an optimal D2-symmetric chiral amidoporphyrin ligand, the Co(II)-based catalytic system is applicable to broad combinations of α-aryldiazomethanes and dehydroaminocarboxylates. In addition to high yields and excellent enantioselectivities under mild conditions, the Co(II)-catalyzed asymmetric cyclopropanation enables direct synthesis of α-amino-β-arylcyclopropanecarboxylates with (Z)-diastereoselectivity, which is different from uncatalyzed thermal reaction. They also present combined computational and experimental studies that shed light on the mechanistic details of the underlying stepwise radical mechanism for the new Co(II)-catalyzed cyclopropanation reaction. Additionally, they show that the resulting enantioenriched (Z)-α-amino-β-arylcyclopropanecarboxylates, as showcased for the efficient synthesis of dipeptides, may serve as useful building blocks for the design and preparation of new peptides with restricted conformations that may have interesting biological properties.
Read the article in Chem.
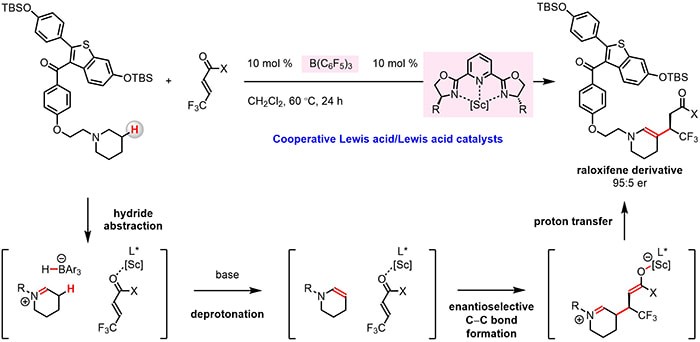
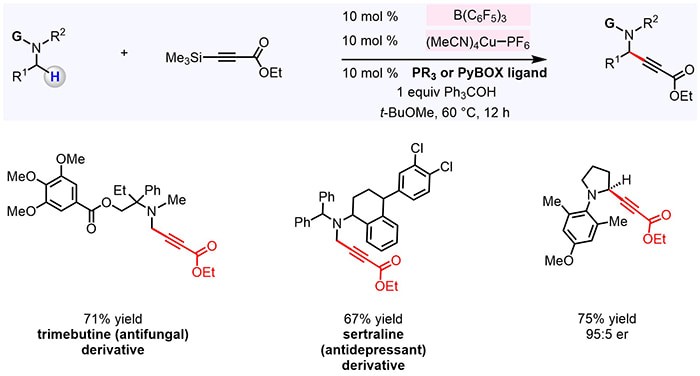
A research team in Professor Masayuki Wasa’s laboratory disclosed an efficient catalytic method for functionalization of β-amino C–H bonds to generate enantioenriched δ-amino carbonyl compounds. They found that by using a blend of B(C6F5)3 and a chiral Sc-based complex, it is possible to convert an N-alkylamine into an enamine and then promote its enantio- and diastereoselective reaction with an α,β-unsaturated compound. The catalyst system was tolerant of a wide variety of Lewis acid sensitive functional units and therefore applicable to late-stage modification of relatively complex (and bioactive) trialkylamine molecules. Mechanistic investigations revealed that the turnover-limiting step is probably isomerization of N-alkylamine-derived iminium ion into an enammonium intermediate. The principles outlined in this work demonstrated that proper combination of an achiral organoborane and an enantiomerically pure organometallic catalyst may be used for chemo- and enantioselective C–H bond functionalization, providing a rational basis for future development of processes for late-stage stereoselective β-functionalization of multifunctional bioactive amines.
Read the article in Journal of the American Chemical Society.
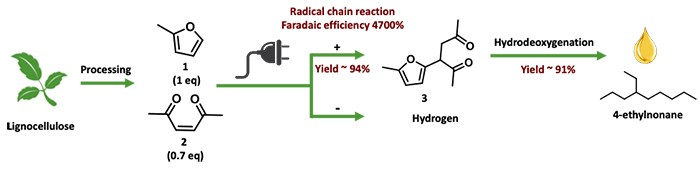
Biomass can serve as an alternative to the ever-depleting fossil fuel resources. The renewable nature of biomass and their carbon dioxide capturing feature promise possibilities of a circular economy. Significant research has been attracted to enable biomass conversion for the production of a variety of important chemicals, especially biomass-derived diesels and jet fuels. In comparison with other chemicals, diesels and jet fuels often require long-chain hydrocarbons that cannot be directly derived from biomass refinement. Coupling reactions that can afford the desired chain lengths from biomass derivatives are often too inefficient or costly or both. The Wang and Niu groups recently developed a unique electrochemical coupling route that may offer a solution to this challenge. Using 2-methyl furan and 3-hexene-2,5-dione, both of which are biomass derivatives, as model compounds, the two teams show that a reactive 2-MF radical can be readily created by electrochemistry. Upon coupling and a single step hydrodeoxygenation to yield branched alkane 4-ethylnonane, the radical is regenerated at the end of the reaction cycle to initiate a new reaction cycle. On average, a single electrochemical electron transfer can produce 47 molecules of the final product, offering an apparent Faradaic efficiency of 4,700%. The only byproduct of the reaction is hydrogen. This new development highlights the power of electrochemically triggered reactions and may find applications in a broad range of reactions.
Read the article in Angewandte Chemie International Edition.
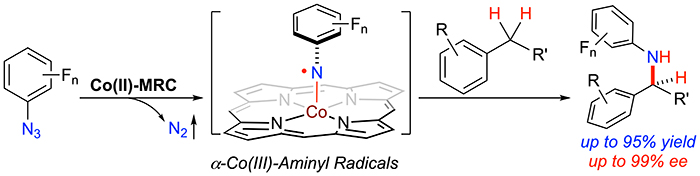
Professor Peter Zhang and coworkers has recently reported a catalytic radical approach that is highly effective for controlling enantioselectivity as well as reactivity of intermolecular C–H amination of carboxylic acid esters with organic azides via Co(II)-based metalloradical catalysis. The key to the success of this radical process lies in the development of the Co(II)-metalloradical catalyst to adopt pocket-like chiral environments through fine-tuning of the remote substituents of the D2-symmetric chiral amidoporphyrin ligands. As a result, it maximizes the cooperative interplay of several noncovalent interactions among the two substrates and the catalyst, including multiple H-bonding and π-stacking interactions as well as van der Waals forces. Together, these attractive weak forces hold the two reacting substrates within the catalyst's pocket in proximity and orient them in proper conformations to facilitate the stereoselective C–N bond formation. This noncovalent interaction strategy presents a solution that may be generally applicable in controlling reactivity and enantioselectivity in intermolecular radical reactions. They show that the Co(II)-catalyzed intermolecular C–H amination, which operates under mild conditions with the C–H substrate as the limiting reagent, exhibits a broad substrate scope with high chemoselectivity, providing effective access to valuable chiral amino acid derivatives with high enantioselectivities. They also present systematic mechanistic studies that shed light into the working details of the underlying stepwise radical pathway for the Co(II)-based C–H amination.
Read the article in Journal of the American Chemical Society.
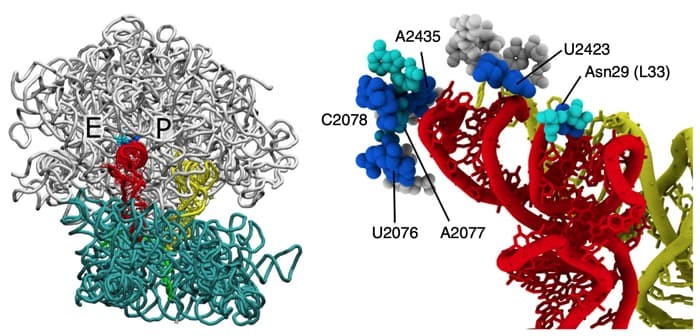
Ribosomes serve as significant biomolecular machines for the synthesis of proteins in living cells, the function of which is achieved by multiple large-scale rearrangements of the ribosome structure and the traverse of tRNA inside. Recently, a team led by Professor Udayan Mohanty, Professor Paul C. Whitford, Dr. Mariana Levi, graduate student Ailun Wang and undergraduate student Kelsey Walak has provided novel insights into the process of P/E hybrid-state formation of tRNA in the ribosome. By performing molecular dynamics simulations using a model that explicitly represents all non-hydrogen atoms, over 120 spontaneous transitions are obtained, where the tRNA molecule displaces between the P and E sites of the large subunit. From the simulation, multiple intermediates of the ribosome with tRNA are predicted which are consistent with experimentally identified features of hybrid formation of tRNA. Energy landscape techniques are applied to identify the molecular factors that govern the biological dynamics of the ribosome, and a six-residue steric gate on the ribosome is reported to control this process and is found limiting the P/E hybrid-state formation of tRNA. Sub-angstrom structural perturbations of the gate-like region can alter the kinetics of the process with an order-of-magnitude change. The precisely defined gate-like residues represent a viable therapeutic target, which may be used to control the functional dynamics of ribosome and tRNA motion. These results establish the relationship between the structure of ribosome and its large-scale conformational dynamics.
Read the article in Nature Communications.
A research team headed by Professor Masayuki Wasa developed an efficient and enantioselective method for activation of a-amino C–H bonds to generate propargyl amines and published this result in the Journal of the American Chemical Society. The group finds that by using a blend of B(C6F5)3 and an organocopper complex, it is possible to generate an iminium from an N-alkylamine and a LnCu–alkynyl complex from an alkynylsilane. The catalyst system tolerates a wide variety of Lewis acid-sensitive functional groups and is therefore applicable to late-stage transformation of a complex (and bioactive) trialkyl amine molecule to its derived propargylamine. Mechanistic investigations indicate that the turnover-limiting step occurs prior to (F5C6)3B-catalyzed C–H abstraction, and that (F5C6)3B-catalyzed C–H abstraction is an irreversible process under the reaction conditions for alkyne incorporation. The principles outlined in their paper demonstrate that proper combination of an achiral organoborane and a chiral organometallic catalyst can be used for chemo- and enantioselective C–H bond activation, providing a rational framework for further development of processes involving the late-stage stereoselective a-functionalization of bioactive amines.
Read the article in Journal of the American Chemical Society.
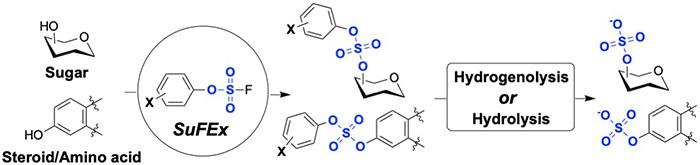
O-Sulfation is an important chemical code widely existing in bioactive molecules and plays important roles in a variety of cell signaling processes. Recently, Assistant Professor Jia Niu, graduate student Chao Liu, and postdoc Cangjie Yang developed a new approach to O-sulfation via Sulfur(VI) Fluoride Exchange (SuFEx) reaction between aryl fluorosulfates and silylated aliphatic alcohols. After a systematic optimization of the electronic properties of aryl fluorosulfates, they demonstrated scalable syntheses of a variety of complex molecules carrying sulfate diesters at various positions, including monosaccharides, disaccharides, amino acid, and steroid. Selective hydrolytic and hydrogenolytic removal of the aryl masking groups from sulfate diesters yielded the corresponding O-sulfated products in excellent yields. This strategy provides a powerful tool for the synthesis of O-sulfated bioactive compounds
Read the article in Angewandte Chemie.
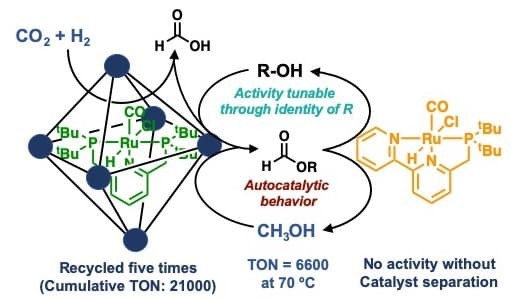
Conversion of a greenhouse gas to a renewable fuel: In this report, a team composed of Professor Jeffery A. Byers, Professor Chia-Kuang (Frank) Tsung, graduate student Thomas Rayder, and undergraduate student Enric Adillon describe a multicomponent catalyst system in which multiple complex catalysts are isolated by a porous crystalline material known as a metal-organic framework (MOF). This system was employed to convert carbon dioxide, a waste product from the combustion of fossil fuels, and hydrogen, which can be obtained from the photocatalytic splitting of water, to methanol. Unlike gaseous hydrogen, methanol is a viable liquid fuel that could be more readily adapted to the current energy infrastructure. As a result of the multicomponent strategy used, the catalyst system demonstrated high activity at lower temperatures than any previously reported catalyst system. Using the MOF host circumvents incompatibility between the two transition metal complexes and allows for catalyst recycling, which is uncommon for other known catalysts for this reaction. The multicomponent catalyst system also uncovered an autocatalytic feature of the reaction that results from the methanol product participating in the second step of the three step reaction sequence. These results illustrate how multiple catalysts sequestered in MOFs enable complex transformations that benefit from a cascade of chemical reactions. It is expected that application of the same strategy to other chemical transformations can lead to new and unexpected reaction manifolds.
Read the article in Chem Journal, Volume: 6, Journal Issue: 7.
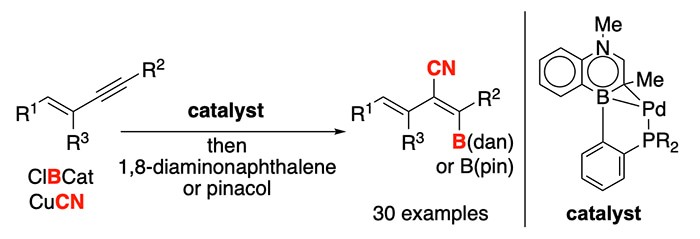
Achieving catalyst-controlled selectivity is an important tenet in catalyst design. A team led by Professor Shih-Yuan Liu and graduate student Yuanzhe Zhang has achieved the first trans-selective cyanoboration reaction of enynes in the presence of a designed catalyst (Angew. Chem. Int. Ed. 2020, 59, 15928-15932. [10.1002/anie.202005882]). This transformation provides access to the important tetra-substituted alkenyl nitrile motif in a site-, regio- and diastereoselective fashion, and its utility is showcased in the synthesis of Satigrel, an anti-platelet aggregating agent. Key to the achievement is the development of the 1,4-azaborine-based biaryl phosphine ligand (Senphos), which engages in a κ2-P-η2-BC binding mode with a palladium (0) source to form the catalytically active species in the reaction process. The catalyst is proposed to engage the substrate cooperatively with chlorocatecholborane in a mechanically unique outer-sphere oxidative addition reaction, which is responsible for the observed selectivity.
Read the article in Angewandte Chemie International Edition.
Suzuki‐Miyaura Cross‐Coupling: In this report, Professor Jeffery A. Byers and graduate students Michael Crockett and Alexander Wong demonstrate the ability to design an iron‐based catalyst supported by β‐diketiminate ligands for reactions between a variety of alkyl halides and unactivated aryl boronic esters. State of the art catalysts for similar transformation rely on toxic nickel-based catalysts and have limited scope with respect to pharmaceutically relevant substrates. This work allows for the use of highly active non-toxic iron-based complexes. High catalyst activity resulted in a broad substrate scope that included tertiary alkyl halides and heteroaromatic boronic esters. Mechanistic experiments revealed that the iron‐based catalyst benefited from the propensity for β‐diketiminate ligands to support low‐coordinate and highly reducing iron amide intermediates, which are very efficient for effecting the transmetalation step required for the Suzuki‐Miyaura cross‐coupling reaction. These findings represent a significant advance in the field of iron-catalyzed coupling reactions and will be a powerful tool for the assembly of biologically active small molecules.
Read the article in Angewandte Chemie, International Edition.
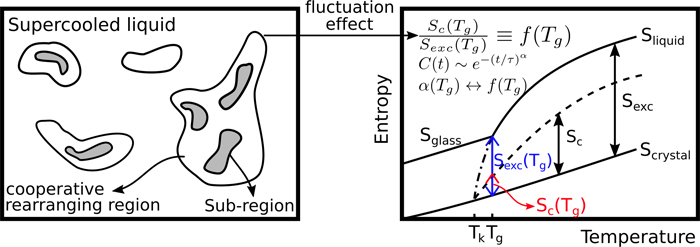
A supercooled liquid is far away from equilibrium. One can measure its properties using variety of experimental probes from dielectric and single molecule spectroscopies to NMR and light scattering. These experiments indicate that there are universal properties such as how diffusion and viscosity behave as a function of temperature and pressure. These universal features have defied statistical mechanical description since configurational entropy that enters kinetic models of supercooled liquids is exceeding difficult to quantify experimentally. Rather, what is measured for such liquids is its excess entropy. The configuration fraction links excess with configurational entropies.
A team lead by Prof. Udayan Mohanty, graduate student Ailun Wang, and postdoc A. Bhattarai have solved a long-standing problem in supercooled liquids. These researchers discovered a deep link between the Kohlrausch stretched exponential parameter that measures long-time decay of time correlation functions of dynamical variables and the configuration fraction. The researchers exploit this connection to predict configuration fraction in glass-forming liquids. The results are in accord with experimental data.
Read the Article in Journal of Physical Chemistry B.
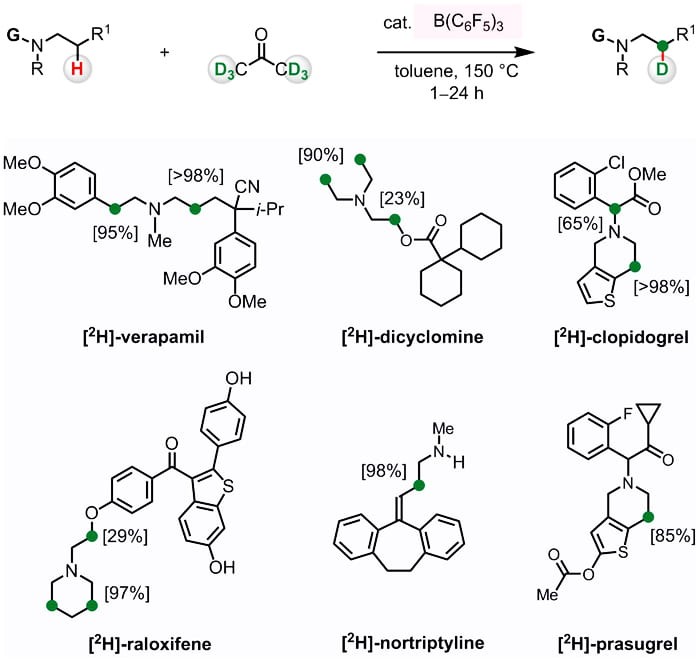
The Wasa Laboratory has designed an efficient and regioselective deuterium labeling of b-amino C–H bonds in various bioactive molecules. By implementing the cooperative action of B(C6F5)3 and N-alkylamine catalyst system, they show that it is possible to convert an N-alkylamine-based pharmaceutical compound to the corresponding enamine, and that the same catalyst system can generate a labeling agent from acetone-d6. The principles outlined in this work entailing conversion of amine containing drugs into enamines and its reaction with in situ generated electrophilic partner, provide a new rational framework for late-stage modification of a drug candidate. Studies along these lines are in progress.
Read the article in Journal of the American Chemical Society.
Under ultrahigh vacuum conditions, a flexible C80H30 warped nanographene synthesized in our laboratory self-assembles on Cu(111) surfaces into the remarkable hexagonal lattice shown. The intermolecular bonding responsible for this pattern depends on both 𝜋-𝜋 and CH•••𝜋 interactions. This self-assembled supramolecular porous network illustrates the potential of negatively curved nanocarbons to form regular homochiral domains. Our discovery resulted from a three-way collaboration involving chemists at Boston College, Nagoya University, and the Swiss Federal Laboratories for Materials Science and Technology.
Read the article in the Journal of the American Chemical Society 2019, 141, 13158-13164.
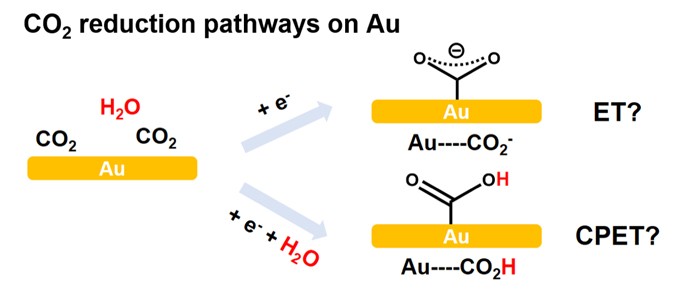
Direct CO2 reduction by methods such as electrochemistry has attracted significant attention not only for its technological implications but also for the scientific challenges it represents. How to steer the reaction toward desired products represents a fundamentally important challenge. Professor Dunwei Wang and his team recently reported a new way of achieving high selectivity in CO2 electrochemical reduction toward CO. The key enabling factor is the unique water-in-salt electrolyte system, where H2O concentration, therefore hydrogen evolution, can be greatly suppressed due to the strong solvation of the high-concentration salt. More importantly, the water-in-salt electrolyte offers an opportunity to tune the H2O concentration for electrokinetic studies of CO2 reduction, a parameter of critical importance to the understanding of the detailed mechanisms but difficult to vary previously. Using Au as a model catalyst platform, a 0 th order dependence of the reaction rate on the H2O concentration was observed, strongly suggesting that electron transfer (ET), rather than concerted proton electron transfer (CPET), from the electrode to the adsorbed CO2 is the rate determining step (RDS). The results shed new light on the mechanistic understanding of CO2 electrochemical reduction. It also sets the stage for future studies on other catalytic systems, as well, which will offer a new dimension to mechanistic studies by tuning H2O concentrations.
Read the article in ACS Central Science
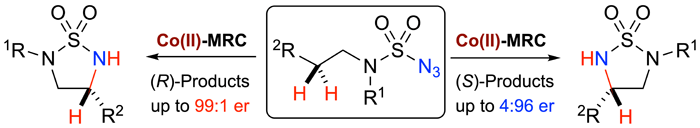
As the latest application of metalloradical catalysis (MRC) for development of stereoselective radical processes, Professor Peter Zhang and coworkers have recently disclosed a new mode of asymmetric induction in catalytic radical C–H amination. Through the employment of bridged chiral porphyrins as the ligands, they have shown that this new mode of asymmetric induction is operational in Co(II)-catalyzed 1,5-C–H radical amination of sulfamoyl azides for formation of strained 5-membered cyclic sulfamides, the enantiocontrol of which has proven difficult. They have further demonstrated that both the degree and sense of the asymmetric induction can be systematically controlled with the use of porphyrin ligands that share the same chiral building block but possess different cavity environment. These studies led to the successful development of a pair of similar Co(II)-based catalysts, which differs only by the length of distal alkyl bridges and the position of the remote non-chiral substitutes in the supporting ligands, for enantiodivergent radical C–H amination through precisely abstracting either pro-(R) or pro-(S) hydrogen in the same methylene unit, followed by stereoretentively homolytic substitution of the resulting radical intermediates at the nitrogen center. The enantiodivergent amination provides a straightforward synthetic tool to access both enantiomers of the desired amination products in high yields with excellent enantioselectivities.
Read the article in Journal of the American Chemical Society.
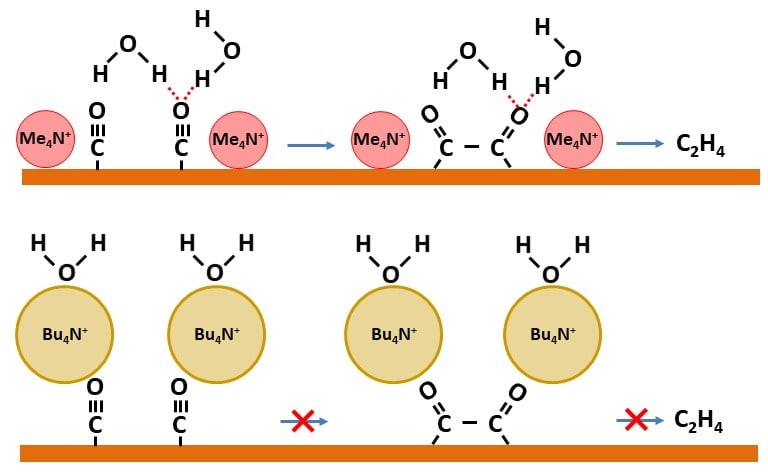
The structure of the electrochemical double layer can greatly affect the reaction selectivity of an electrocatalytic process. Many electrocatalytic processes of current interest involve the transfer of multiple protons and electrons. Examples include the electrocatalytic reductions of nitrogen, nitrate, and carbon dioxide. These processes often exhibit poor selectivity for desired products. To improve the reaction selectivity, it is essential to understand how the interfacial environment at the electrode/electrolyte contact controls the reaction path. This understanding has not been sufficiently developed to date. In their latest advance, Professor Matthias Waegele and his graduate students Jingyi Li, Xiang Li, and Charuni Gunathunge show that hydrogen bonding of interfacial water to a critical surface-intermediate is essential for the electrocatalytic reduction of carbon monoxide to ethylene, a prototypical multi-proton/electron transfer process. Using a series of quaternary alkyl ammonium cations, the team has systematically tuned the structure of the electrochemical double layer. With differential electrochemical mass spectrometry (DEMS), they show that ethylene is produced in the presence of methyl4N+ and ethyl4N+ , whereas it is not formed in propyl4N+- and butyl4N+- containing electrolytes. To understand these findings, the team probed the structure of the electrochemical double layer with surface-enhanced infrared absorption spectroscopy (SEIRAS). Using this technique, they demonstrate that an intermolecular interaction of interfacial water with surface-adsorbed CO is disrupted in the presence of the two larger, more hydrophobiccations (propyl 4 N+ , butyl4N+ ). This observation suggests that this interaction is critical for the formation of ethylene. This mechanistic insight is expected to aid the development of electrocatalytic interfaces that exhibit high product selectivity.
Read the article in Proceedings of the National Academy of Sciences
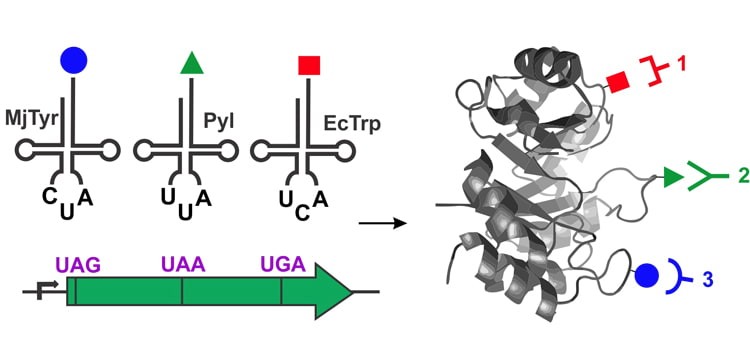
The ability to precisely label proteins with multiple distinct entities at predefined locations is highly desirable for numerous cross-disciplinary applications. Co-translational site-specific incorporation of noncanonical amino acids (ncAA) enables precise placement of uniquely reactive chemical functionalities into proteins, which can be subsequently labeled using chemoselective bioconjugation reactions. A team led by Associate Professor Abhishek Chatterjee and graduate students James Italia has developed a strategy for site-specifically incorporating up to three distinct ncAAs into recombinant proteins. Furthermore, the team used this strategy to site-specifically incorporate three distinct functionalities into a protein (an azide, a cyclopropene, and a 5-hydroxyindole), which can be independently labeled with high selectivity by three different mutually orthogonal bioconjugation reactions. The ability to site-selectively introduce three different labels into one protein will facilitate many powerful applications, including the attachment of different probes for sophisticated biophysical studies, and the synthesis of advanced protein-based therapeutics and diagnostics.
Read the article in the Journal of the American Chemical Society.
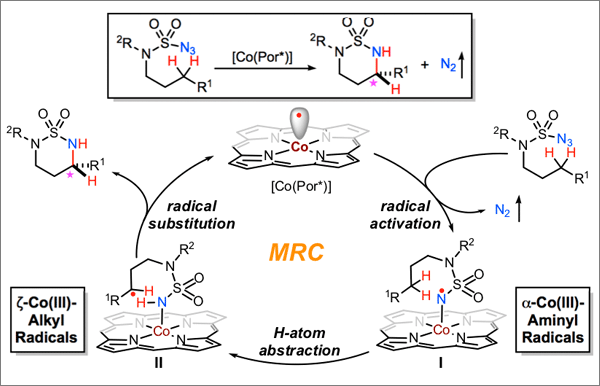
As the latest application of metalloradical catalysis (MRC) for development of stereoselective radical processes, Professor Peter Zhang and coworkers have recently reported the first asymmetric catalytic system via Co(II)-based metalloradical catalysis for highly enantioselective 1,6-C(sp3)–H amination of sulfamoyl azides under neutral and non-oxidative conditions. They describe that the Co(II) complex of a structure-optimized D2-symmetric chiral amidoporphyrin can efficiently catalyze asymmetric 1,6-C(sp3)–H radical amination even at room temperature, forming the six-membered cyclic sulfamides in high yields with excellent enantioselectivities. In addition to benzylic C(sp3)–H bonds, they demonstrated that the Co(II)-based metalloradical system is capable of aminating a broad spectrum of substrates, including allylic, propargylic, non-activated, and even electron-deficient C(sp3)–H bonds. Additional features of the Co(II)-catalyzed C–H amination include a remarkable degree of functional group tolerance and an unusual level of chemoselectivity. The resulting optically active six-membered cyclic sulfamides, which are interesting N-heterocycles in their own right, can be easily converted to synthetically useful 1,3-diamines with full retention of the original enantiopurity.
Read the article in Angewandte Chemie International Edition.
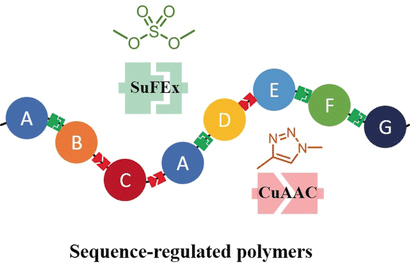
Achieving control over the sequence of synthetic polymers is widely considered as a “holy grail” in the field of polymer chemistry, enabling promising applications in catalysis, molecular recognition, data storage, and biomimicry. A team led by assistant professor Jia Niu and also including postdoc Cangjie Yang and BC undergraduate James Flynn developed a facile approach to generate sequence-regulated synthetic polymers. Their approach is based on the combination of two orthogonal click chemistries, the CuAAC and the SuFEx reactions, leading to polydispersed sequence-controlled polymers and monodispersed sequence-defined oligomers in high yield and sequence complexity. This work represents the first systematic study on the orthogonality of the CuAAC and SuFEx click reactions and demonstrated how the orthogonal click reactions can be used to facilely access a broad range of sequence-regulated polymers.
This work was recently published in Angewandte Chemie International Edition.
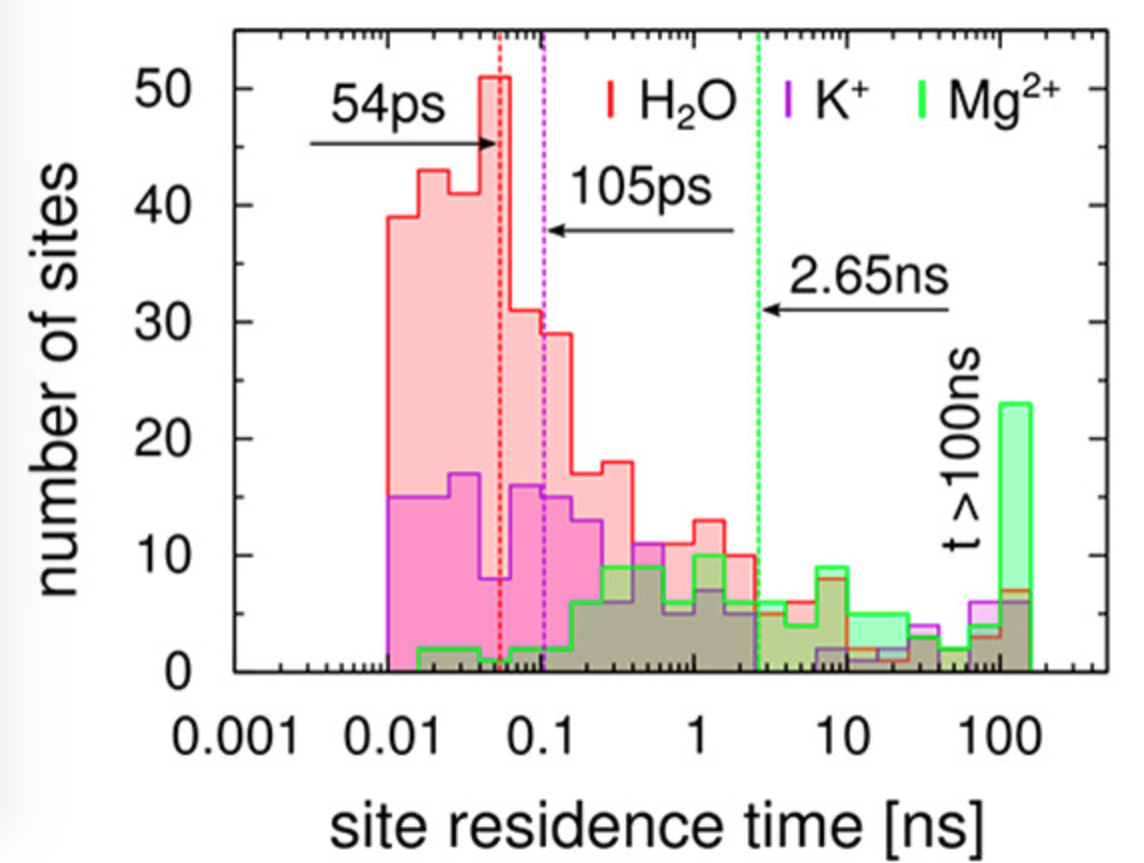
The importance of Mg2+ counterions for the stability of compact RNAs is widely recognized, and several recent studies have aimed to characterize Mg2+ binding to folded RNA molecules. But the influence of the solution environment on RNA dynamics that is the basis of its plasticity is not yet understood due to difficulties in resolving motional modes that span a wide range of spatial and temporal scales. A team lead by Prof. Udayan Mohanty, Prof. Jose N Onuchic, Dr. Heiko Lammert and graduate student Ailun Wang have provided new insights into the characteristics of the strong interactions between a folded 58 nucleotide ribosomal RNA fragment and the ionic environment that results in RNA's ability to rapidly change its conformation.
The researchers show that the 58mer rRNA in solution forms a complex polymer, composed of RNA lubricated by a fluctuating network of associated ions and water. The team determine stabilizing interactions and coupled dynamics for all solvent species, which enhance the RNA’s capacity for rapid conformational changes. Dynamics is not simply determined by the RNA but is shaped jointly by thisintegrated super-molecular system.
The researchers determine the spatial and the temporal distribution of residence times in all the association sites for ions and solvating water near the RNA from atomic resolution explicit solvent molecular dynamics simulations. The long-lived outer-sphere association sites anchor the local ionic RNA environments for long times against fluctuations. Dynamical couplings join the RNA with the divalent and monovalent metal cations and water that form its solvation sphere into shared local and even global modes.
A significance of this work is that the researchers provide detailed insights into collective and local dynamical modes of the 58-mer rRNA fragment that form the basis for its structural adaptation in solution environment. This work underscores the importance of discrete ion effects of the outer-sphere on RNA dynamics.
Read the Article in Journal of Physical Chemistry B, DOI 10.1021/acs.jpcb.8b06874
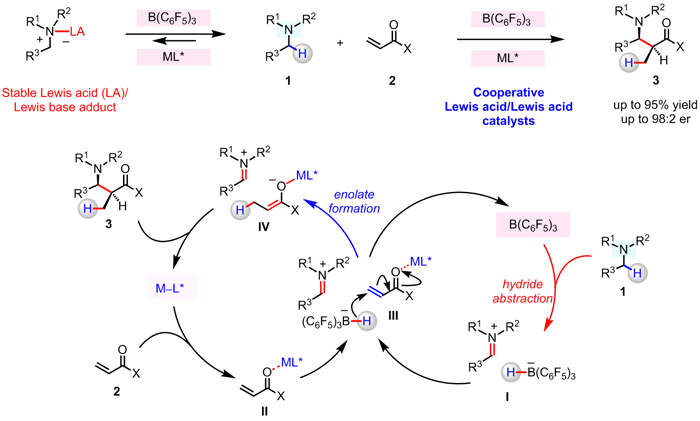
Professor Masayuki Wasa along with postdoctoral fellow Ming Shang, graduate students Jessica Chan, Min Cao, Yejin Chang and Qifan Wang, as well as undergraduate Brennan Cook and research fellow Sebastian Torker have reported a strategy for enantioselective union of N-alkylamines and α,β-unsaturated compounds, performed under redox-neutral conditions, and promoted through concerted action of seemingly competitive Lewis acids, B(C6F5)3 and a chiral Mg–PyBOX complex. By employing this approach to reaction design, a wide variety of β-amino carbonyl compounds may be synthesized, with complete atom economy, through stereoselective reaction of an in situ generated enantiomerically enriched Mg-enolate and an appropriate electrophile.
This work was published in the Journal of the American Chemical Society [doi:10.1021/jacs.8b06699].
Taking water and ripping it apart into hydrogen and oxygen could form the basis of artificial photosynthetic devices that could ultimately power homes and businesses. However, catalysts, including those used to "split" water, have either worked well but are expensive and unstable, or are affordable and stable, but don't work as well. Now, Prof. Dunwei Wang’s team and collaborators report in ACS Central Science a new catalyst that is really the best of both worlds.
Identifying ideal materials that can split water is a long-standing problem in renewable energy storage. Catalysts, which help reactions occur, are often used in this process. "Homogeneous" ones dissolve into the reaction solution and are usually active and selective. However, they don't work well in some applications because they are unstable and expensive. In contrast, "heterogeneous" catalysts are solids that are stable, recyclable and convenient to work with, but they are usually not very active or selective. The Boston College-led team proposed they could get closer to the ideal catalyst by producing a hybrid material.
The researchers developed a new hybrid catalyst made of iridium dinuclear heterogeneous catalysts (DHCs) attached to a tungsten oxide substrate. They found that attaching the ends of the DHC molecules -- instead of the sides -- allowed the catalyst to perform optimally. The researchers suggest that this first-of-its-kind material could be an important step toward alternative solar energy storage or artificial photosynthesis.
Link to paper: https://pubs.acs.org/doi/10.1021/acscentsci.8b00335
As a powerful approach to introduce functional groups into the polymer backbone, existing ring-opening polymerization techniques nevertheless have limited utility for macrocyclic monomers due to the lack of ring strain. A team led by assistant professor Jia Niu and also including postdoc Hanchu Huang and BC undergraduates Bohan Sun and Yingzi Huang aimed to overcome this challenge. They designed an allyl alkylsulfone-based ring-opening trigger that can undergo a radical cascade reaction to extrude sulfur dioxide and generate an alkyl radical for controlled polymerization. They performed a systematic study correlating reaction conditions with polymer molecular weight and molecular weight distribution to achieve excellent control. The versatility of this radical cascade-triggered ring-opening polymerization approach was further demonstrated through the first radical block copolymerization of macrocyclic monomers, and the incorporation of degradable functionality into the polymer backbone. This work was recently published in Journal of the American Chemical Society (J. Am. Chem. Soc., 2018, 140 (33), pp 10383–10384).
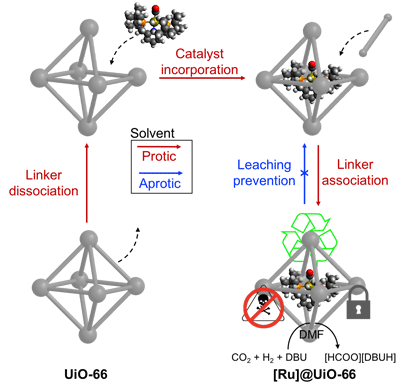
Conversion of CO 2 into useful liquid fuels is an attractive way to address dwindling fossil fuel resources. Heterogeneous catalysts used for these processes either suffer from harsh reaction conditions or poor product selectivities. Homogeneous catalysts suffer from poor recyclability and are prone to deactivation from poisons in the gaseous feed. Collaborative work from Professors Jeffery A. Byers and Frank Tsung spearheaded by their joint graduate students Zhehui Li and Tom Rayder has recently made it possible to convert a homogeneous catalyst used for CO 2 hydrogenation into a catalyst construct that demonstrates behavior hybrid between homogeneous and heterogeneous catalysts. In a report in the Journal of the American Chemical Society, the team capitalizes on a solvent dependent dissociative ligand exchange processes that take place in porous three-dimensional crystals known as metal-organic frameworks (MOFs) to encapsulate a molecular transition metal complex that is capable of catalytically converting CO 2 to formate. Unlike its homogeneous counterpart, the “aperture-opening encapsulation” resulted in isolation of catalytically active sites that made the hybrid catalyst more easily recyclable and less prone to catalyst poisoning. The success of this process opens the door for the development of a variety of homogeneous/heterogeneous hybrid materials through variation in both the homogeneous catalyst and porous framework. This work was recently published in Journal of the American Chemical Society.
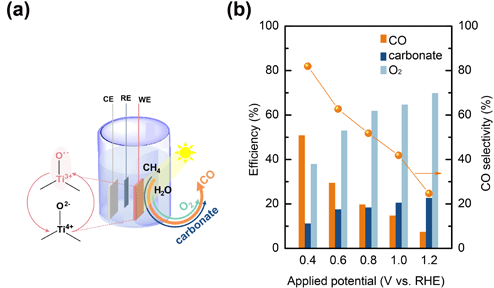
Selective oxidation of CH4 is an important process that produces high-value industrial chemicals such as CO or CH3OH. Photocatalytic approach to this process holds great promise for practical applications but was poorly studied. Recently Prof. Wang and Prof. Waegele reported a significant step forward in this area. Graduate students Wei Li and Da He, together with other collaborators at UC Riverside and Yale University, successfully demonstrated selective CH4 photo-oxidation with a high yield of CO production. The key to their success was controlled activation of CH4 without over-oxidizing it all the way to CO2. Detailed experimental and computational investigations highlight the importance of synergistic effects by adjacent Ti sites on the TiO2 catalyst to the unique selectivity. The study not only presents a new route to selective CH4 oxidation, but also shed important light on other heterogeneous catalysis such as CO2 reduction.
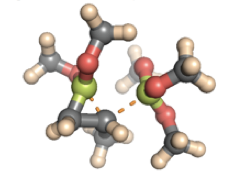
By means of quantum chemical calculations Professor Haeffner has examined the molecular mechanisms underlying a novel transition-metal- free diboration reaction of olefins. The computational study shows that the reaction can proceed via four distinctively different non-concerted pathways A, B, C and D depending on which diboration reagent is being used. These new insights may guide further method development of this important chemical transformation. Shown below are the energetically favored transition structures along these pathways controlling the stereochemistry of the highly stereoselective SYN-diboration reaction between B 2 pin 2 and propene.
Read the article in Computational and Theoretical Chemistry.
![The Wang group has led an international collaboration in the development of a new heterogeneous catalyst for solar water splitting applications. The catalyst consists of two Ir atoms as the active site. It represents one of the first examples of such catalysts with well- defined atomic structures. Most other high-performance heterogeneous catalysts are poorly defined in their active centers. To correct the deficiency, graduate student Yanyan Zhao from the Wang group started from an Ir molecular catalyst ([Ir(pyalc)(H 2 O) 2 (μ-O)] 2 2+ homo-dimer) and immobilize it onto Fe 2 O 3 surface. A subsequent photochemical treatment was found to strip off all organic ligands to yield the Ir-O- Ir dimer that are stabilized by only H 2 O, OH - and surface O. To study the structures, the authors employed a suite of characterization tools, including aberration-corrected TEM, X-ray absorption spectroscopy and diffuse reflectance infrared Fourier transform spectroscopy. The unique structure was unambiguously confirmed. Importantly, the Ir-O- Ir catalyst exhibited high activity toward solar water oxidation. The well-defined structure allowed the authors to propose the reaction mechanisms, which were confirmed by DFT calculations. A new door to heterogeneous catalyst preparation and study that can directly benefit from knowledge gained on homogeneous catalysis is opened up. The study also involved Yale University, Tufts University, University of California Irvine, the Advanced Light Source, Tsinghua University and Nanjing University.](/content/bc-web/schools/morrissey/departments/chemistry/research/_jcr_content/bottompar/bc_padded_section/par/bc_image_content_44/image.img.png/1627508707839.png)
A High Throughput Platform for Developing Targeted Antibiotics
The use of broad-spectrum antibiotics is less ideal as it unavoidably accelerates bacterial resistance to antibiotics and may cause damage to human microbiota as well. Ideally, one would like to treat infection with targeted antibiotics that specifically eradicate the infection-causing pathogen. Progress toward this end has been hampered by the difficulty of developing targeting motifs for various bacterial species. Reporting in Journal of the American Chemical Society, a joint team of Associate Professors Jianmin Gao (Chemistry) and Tim van Opijnen (Biology) has developed a high throughput platform to enable quick identification of molecular probes for a bacterium of interest. This new platform builds on phage display, a strategy to create and screen peptide libraries. Although powerful, phage display has been largely limited to peptides of natural amino acids. In the current publication, the authors sought to expand the chemical space of phage display by incorporating dynamic covalent binding motifs. Screening of the chemically modified phage library has revealed potent and selective probes for a methicillin-resistant Staphylococcus aureus and a colistin-resistant Acinetobacter baumannii, both important bacterial pathogens. The team further demonstrates that targeted antibiotics can be created by linking a generic toxin to these bacterium-targeting molecules. This approach should be applicable to a wide range of bacterial pathogens, paving the way to facile development of targeted antibiotics.
Kelly A. McCarthy, Michael A. Kelly, Kaicheng Li, Samantha Cambray, Azade S. Hosseini, Tim van Opijnen, and Jianmin Gao*, “Phage display of dynamic covalent binding motifs enables facile development of targeted antibiotics,” J. Am. Chem. Soc. DOI: 10.1021/jacs.8b02461.
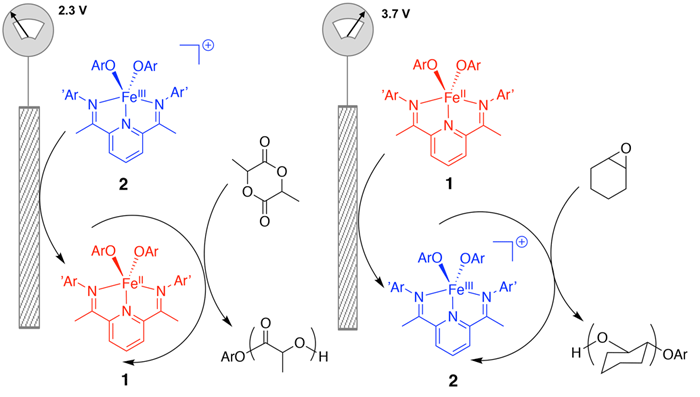
Switchable Catalysis: In this report, Professor Jeffery A. Byers, Professor Dunwei Wang, and graduate students Miao Qi and Qi Dong demonstrate how the reactivity of an iron-based polymerization catalyst can be altered in situ through application of an electrical current. By using electrical potential as a toggle switch, lactide and cyclohexene oxide polymerizations could be selectively polymerized from a reaction vessel that contained a mixture of both monomers. As a result, block copolymers could be synthesized, in which the sequence of such copolymer is dictated by the applied potential. These findings mark a significant advance forward towards the long-standing goal for polymer chemists to create programmable polymer sequences that resemble natural biopolymers, such as proteins, complex carbohydrates, and DNA.
Read the article in Journal of the American Chemical Society, DOI: 10.1021/jacs.8b02171
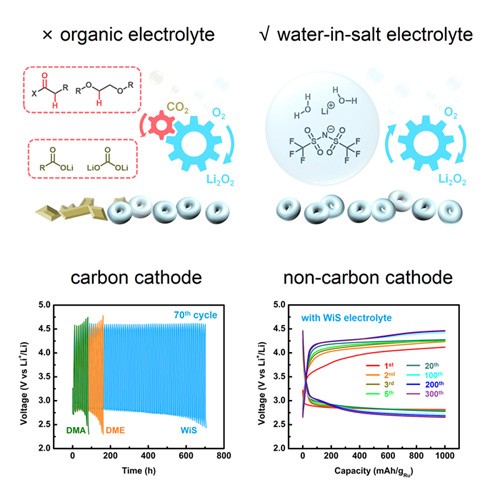
As an electrochemical energy storage technology that holds the highest theoretical energy density, Li-O 2 batteries have been studied for over two decades. Its reported performance, however, remains much lower than expected. An important reason for the slow progress is the lack of stable electrolytes. Because of the high reactivity of oxygen species in the system, no known organic electrolytes meet the stability requirements. The search for a suitable electrolyte system remains an outstanding challenge in Li-O 2 battery research. To correct the deficiency, Professor Dunwei Wang and his team show that the issue can be solved with the use of a water-in-salt electrolyte system that involves no organic solvents. In essence, the electrolyte consists of super-concentrated LiTFSI (21 molality of lithium bis(trifluoromethanesulfonyl)imide), in which H 2 O molecules are locked to the ions and exhibit little reactivity toward the desired discharge product Li 2 O 2 or other oxygen species. Both qualitative and quantitative product analyses support that no measurable by-products form in the WiS system. With a carbon cathode, greatly improved cyclability of over 70 cycles can be obtained with WiS electrolyte in comparison with organic electrolytes. When the carbon cathode is replaced with a stable carbon-free material Ru/TiSi 2 , up to 300 cycles of Li-O 2 battery operations are obtained. This result sets a new benchmark in Li-O 2 battery performance with quantitative product detection. It also sets the stage for future optimizations to realize the full potential held by Li-O 2 batteries as a stable, high-capacity electrochemical energy storage technology.
Featured in Chem, Volume 4.
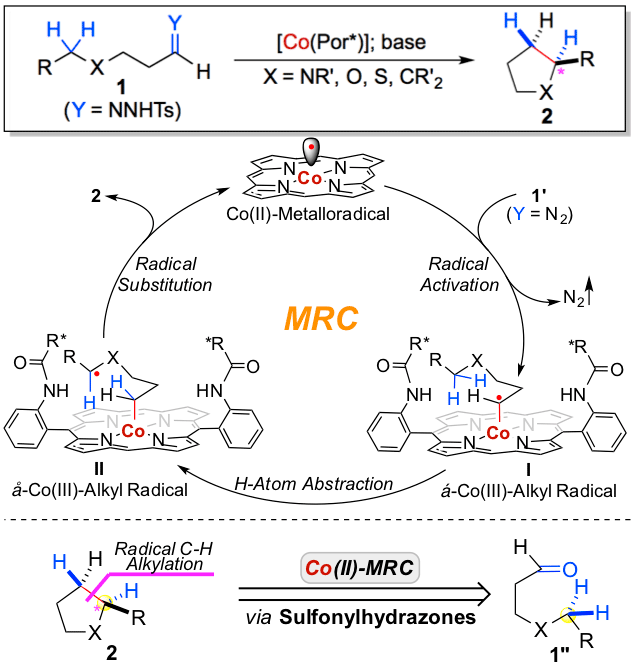
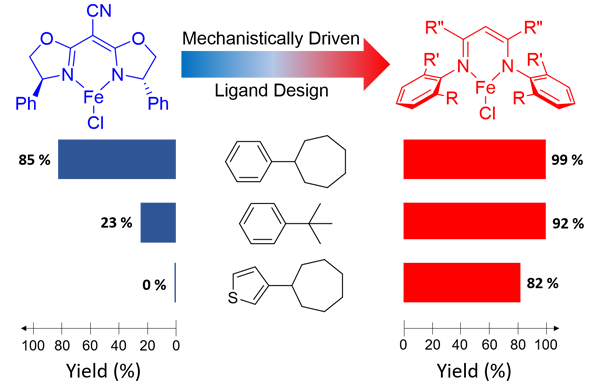
Radical cyclization represents a powerful strategy for construction of ring structures. Traditional radical cyclization, which is based on radical addition as the key step, necessitates the use of unsaturated substrates. As the latest synthetic application of metalloradical catalysis (MRC), the research laboratory of Professor Peter Zhang, led by graduate students Yong Wang and Xin Wen, has demonstrated a different mode of radical cyclization that can employ saturated C–H substrates. They have developed a new Co(II)-based metalloradical system that can catalytically activate aliphatic diazo compounds for enantioselective radical alkylation of various C(sp 3 )–H bonds, allowing for efficient construction of chiral pyrrolidines and other valuable 5-membered compounds. In addition to chemoselectivity and regioselectivity, this new radical cyclization features functional group tolerance and heteroarene compatibility. This alternative strategy of radical cyclization provides a new retrosynthetic paradigm to prepare five-membered cyclic molecules from readily available linear aldehydes through the union of C–H and C=O elements for C–C bond formation.
Read the article in Journal of the American Chemical Society.
![The Wang group has led an international collaboration in the development of a new heterogeneous catalyst for solar water splitting applications. The catalyst consists of two Ir atoms as the active site. It represents one of the first examples of such catalysts with well- defined atomic structures. Most other high-performance heterogeneous catalysts are poorly defined in their active centers. To correct the deficiency, graduate student Yanyan Zhao from the Wang group started from an Ir molecular catalyst ([Ir(pyalc)(H 2 O) 2 (μ-O)] 2 2+ homo-dimer) and immobilize it onto Fe 2 O 3 surface. A subsequent photochemical treatment was found to strip off all organic ligands to yield the Ir-O- Ir dimer that are stabilized by only H 2 O, OH - and surface O. To study the structures, the authors employed a suite of characterization tools, including aberration-corrected TEM, X-ray absorption spectroscopy and diffuse reflectance infrared Fourier transform spectroscopy. The unique structure was unambiguously confirmed. Importantly, the Ir-O- Ir catalyst exhibited high activity toward solar water oxidation. The well-defined structure allowed the authors to propose the reaction mechanisms, which were confirmed by DFT calculations. A new door to heterogeneous catalyst preparation and study that can directly benefit from knowledge gained on homogeneous catalysis is opened up. The study also involved Yale University, Tufts University, University of California Irvine, the Advanced Light Source, Tsinghua University and Nanjing University.](/content/bc-web/schools/morrissey/departments/chemistry/research/_jcr_content/bottompar/bc_padded_section/par/bc_image_content_42/image.img.jpg/1627508726103.jpg)
Natural products are an important proving ground to examine the utility of new chemical methods and to inspire the development of new transformations. In this context, the Morken Laboratory has engaged in the construction of the natural product arenolide, a modestly cytotoxic compound isolated from a marine sponge in Palau. Graduate students Xun Liu (now a Ph.D. research scientist at Incyte Pharmaceuticals), Scott Mlynarski (currently at Astra Zeneca) along with postdoc Chunrui Sun (now at Merck) completed a convergent synthesis of candidate stereoisomers of the natural product by employing recently developed catalytic boron-based reactions from the Morken laboratory. Comparison of the spectral data for candidate structures with that reported for the authentic natural product revealed the likely stereostructure of the natural compound.
The manuscript was published in Organic Letters (2018, 20, 1898-1901; doi: 10.1021/acs.orglett.8b00439)
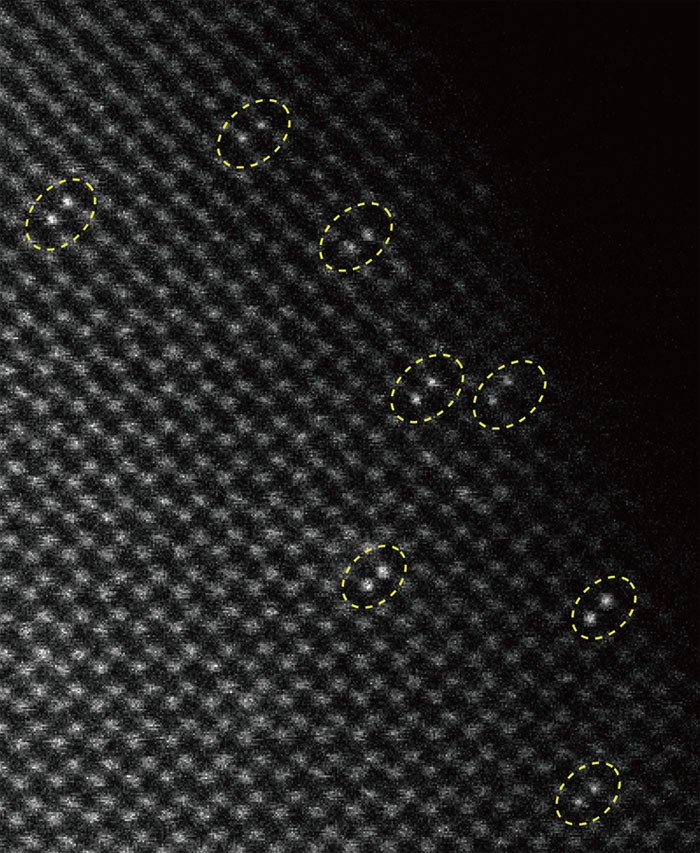
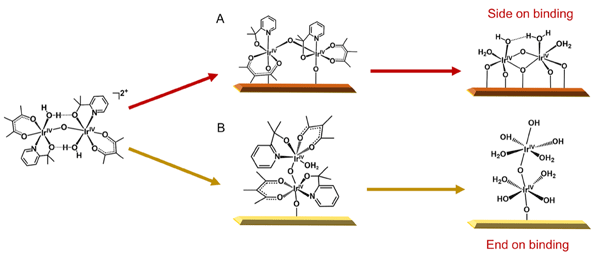
The Wang group has led an international collaboration in the development of a new heterogeneous catalyst for solar water splitting applications. The catalyst consists of two Ir atoms as the active site. It represents one of the first examples of such catalysts with well-defined atomic structures. Most other high-performance heterogeneous catalysts are poorly defined in their active centers. To correct the deficiency, graduate student Yanyan Zhao from the Wang group started from an Ir molecular catalyst ([Ir(pyalc)(H2O)2(μ-O)]22+ homo-dimer) and immobilize it onto Fe2O3 surface. A subsequent photochemical treatment was found to strip off all organic ligands to yield the Ir-O- Ir dimer that are stabilized by only H2O, OH - and surface O. To study the structures, the authors employed a suite of characterization tools, including aberration-corrected TEM, X-ray absorption spectroscopy and diffuse reflectance infrared Fourier transform spectroscopy. The unique structure was unambiguously confirmed. Importantly, the Ir-O- Ir catalyst exhibited high activity toward solar water oxidation. The well-defined structure allowed the authors to propose the reaction mechanisms, which were confirmed by DFT calculations. A new door to heterogeneous catalyst preparation and study that can directly benefit from knowledge gained on homogeneous catalysis is opened up. The study also involved Yale University, Tufts University, University of California Irvine, the Advanced Light Source, Tsinghua University and Nanjing University.
![The Wang group has led an international collaboration in the development of a new heterogeneous catalyst for solar water splitting applications. The catalyst consists of two Ir atoms as the active site. It represents one of the first examples of such catalysts with well- defined atomic structures. Most other high-performance heterogeneous catalysts are poorly defined in their active centers. To correct the deficiency, graduate student Yanyan Zhao from the Wang group started from an Ir molecular catalyst ([Ir(pyalc)(H 2 O) 2 (μ-O)] 2 2+ homo-dimer) and immobilize it onto Fe 2 O 3 surface. A subsequent photochemical treatment was found to strip off all organic ligands to yield the Ir-O- Ir dimer that are stabilized by only H 2 O, OH - and surface O. To study the structures, the authors employed a suite of characterization tools, including aberration-corrected TEM, X-ray absorption spectroscopy and diffuse reflectance infrared Fourier transform spectroscopy. The unique structure was unambiguously confirmed. Importantly, the Ir-O- Ir catalyst exhibited high activity toward solar water oxidation. The well-defined structure allowed the authors to propose the reaction mechanisms, which were confirmed by DFT calculations. A new door to heterogeneous catalyst preparation and study that can directly benefit from knowledge gained on homogeneous catalysis is opened up. The study also involved Yale University, Tufts University, University of California Irvine, the Advanced Light Source, Tsinghua University and Nanjing University.](/content/bc-web/schools/morrissey/departments/chemistry/research/_jcr_content/bottompar/bc_padded_section/par/bc_image_content_41/image.img.png/1627508574909.png)